CBSE Class 10 Science MCQs for Term 1
Class 10 Science MCQ: CBSE has begun with the conduction of the Class 10 Term-1 exam and has scheduled the Class 10 Science Exam for 02nd December 2021. With only 2 days left for the exam, the students must practice as much as Class 10 Science MCQs to boost up their preparation and confidence to appear in the real Term-1 Exam. Solving Science MCQ Class 10 for Term-1 will help the students to have an idea of how the questions can be asked and how much time they should give to each question.
Science MCQs for Class 10 Term-1
In the below article, we have provided you with some important questions as per the new exam pattern Do go through these Class 10 Science MCQs for Term-1 and excel your preparation done so far. Till now, CBSE has conducted one-time descriptive exams and this is the first time CBSE has divided papers into Term-1 & Term-2. There will be Multiple-Choice Questions in Term-1 and helping you to better understand the pattern, practice these Class 10 Science MCQ as per CBSE Term-1 Syllabus.
Science Class 10 Term-1 Answer Key 2021 Out- Download Link
CBSE Class 10th Science MCQ Paper
Practice below given Science MCQ Class 10 Questions for Term-1 Exam to be held on 02nd December 2021 and at the end of the article, we have listed the answers to each question. But we advised you to solve these questions first and then reach out to the answer section.
CBSE Class 10 Science MCQs for Term 1
Q1. Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of Potassium Iodide solution to it. What would she observe?
Option A The solution turned red.
Option B Yellow precipitate was formed
Option C White precipitate was formed
Option D The reaction mixture became hot
Q2. Which of the following correctly represents a balanced chemical equation?
Option A Fe(s) + 4H2O(g) → Fe3O4 (s) + 4H2(g)
Option B 3Fe(s) + 4H2O(g) → Fe3O4 (s) + 4H2(g)
Option C 3Fe(s) + H2O(g) → Fe3O4 (s) + H2(g)
Option D 3Fe(s) + 4H2O(g) → Fe3O4 (s) + H2(g)
Q3. In the reaction of iron with copper sulphate solution: CuSO4 + Fe ---> Cu + FeSO4
Which option in the given table correctly represents the substance oxidised and the reducing agent?
| Option | Substance Oxidized | Reducing Agent |
| Option A | Fe | Fe |
| Option B | Fe | FeSO4 |
| Option C | Cu | Fe |
| Option D | CuSO4 | Fe |
Q4. The chemical reaction between copper and oxygen can be categorized as:
Option A Displacement reaction
Option B Decomposition reaction
Option C Combination reaction
Option D Double displacement reaction
Q5. Which of the given options correctly represents the Parent acid and base of Calcium Carbonate?
| Option | Parent Acid | Parent Base |
| Option A | HCl | NaOH |
| Option B | H2CO3 | Ca(OH)2 |
| Option C | H3PO3 | CaSO4 |
| Option D | H2SO | CaSO4 |
Q6. Before burning in air, the magnesium ribbon is cleaned by rubbing with sandpaper to:
Option A Make the ribbon surface shinier
Option B Remove the layer of magnesium oxide from the ribbon surface
Option C Remove the layer of magnesium carbonate from the ribbon surface
Option D Remove the moisture from the ribbon surface
Q7. A chemical reaction does not involve:
Option A Formation of new substances having entirely different properties than that of the reactants
Option B Breaking of old chemical bonds and formation of new chemical bonds
Option C Rearrangement of the atoms of reactants to form new products
Option D Changing the atoms of one element into those of another element to form new products.
Q8. How will you protect yourself from the heat generated while diluting a concentrated acid?
Option A By adding acid to water with constant stirring.
Option B By adding water to acid with constant stirring.
Option C By adding water to acid followed by the base.
Option D By adding base to acid with constant stirring.
Q9. Why is it important to balance a skeletal chemical equation?
Option A To verify the law of conservation of energy.
Option B To verify the law of constant proportion.
Option C To verify the law of conservation of mass.
Option D To verify the l0aw of conservation of momentum.
Q10. Carefully study the diagram of the human respiratory system with labels A, B, C and D. Select the option which gives correct identification and main function and /or characteristic.

Option A (i) Trachea: It is supported by bony rings for conducting inspired air.
Option B (ii) Ribs: When we breathe out, ribs are lifted.
Option C (iii) Alveoli: Thin-walled sac-like structures for exchange of gases.
Option D (iv) Diaphragm: It is pulled up when we breathe in.
Q11. Identify the option that indicates the correct enzyme that is secreted in locations A, B and C.
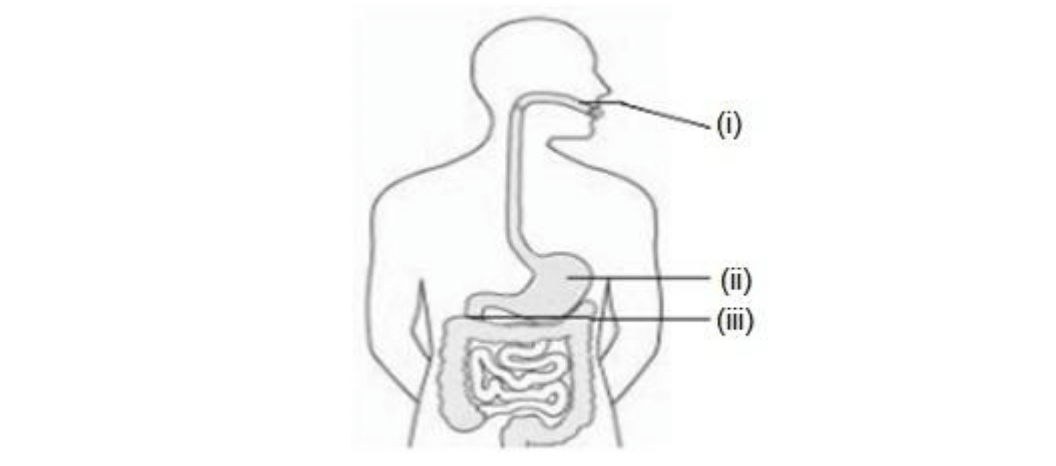
Option A (i)-lipase, (ii)-trypsin, (iii)-pepsin
Option B (i)-amylase, (ii)-pepsin, (iii)-trypsin
Option C (i)-trypsin, (ii)-amylase, (iii)-carboxylase
Option D (i)-permease, (ii)-carboxylase, (iii)-oxidase
Q12. Opening and closing of stomatal pore depends on:
Option A Atmospheric temperature
Option B Oxygen concentration around stomata
Option C Carbon dioxide concentration around stomata
Option D Water content in the guard cells
Q13. The graph given below depicts a neutralization reaction (acid + alkali → salt + water). The pH of a solution changes as we add excess of acid to an alkali.
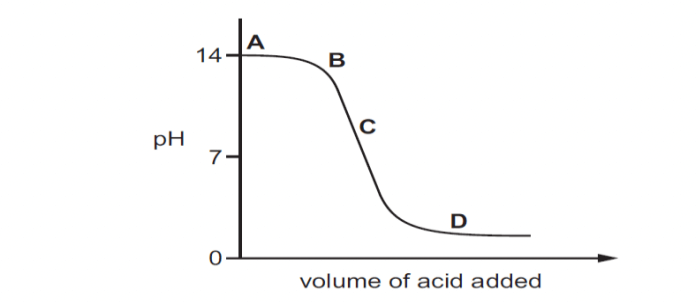
Which letter denotes the area of the graph where both acid and salt are present?
Option A A
Option B B
Option C C
Option D D
Q14. Which of the following mirror is used by a dentist to examine a small cavity in a patient’s teeth?
Option A Convex mirror
Option B Plane mirror
Option C Concave mirror
Option D Any spherical mirror
Q15. Which of the following can make a parallel beam of light when light from a point source is incident on it?
Option A Concave mirror as well as a convex lens.
Option B Convex mirror as well as a concave lens.
Option C Two plane mirrors placed at 90° to each other.
Option D Concave mirror as well as a concave lens.
Q16. Consider these indices of refraction: glass: 1.52; air: 1.0003; water: 1.333. Based on the refractive indices of three materials, arrange the speed of light through them in decreasing order.
Option A The speed of light in water >> the speed of light in air >> the speed of light in glass.
Option B The speed of light in glass >> the speed of light in water >> the speed of light in air.
Option C The speed of light in air >> the speed of light in water >> the speed of light in glass.
Option D The speed of light in glass >> the speed of light in air >> the speed of light in water.
Q17. The figure given below shows a schematic plan of blood circulation in humans with labels (i) to (iv). Identify the correct label with its functions?
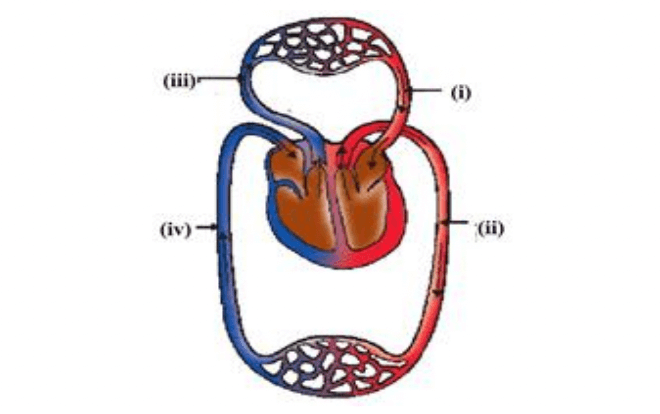
Option A (i) Pulmonary vein - takes impure blood from the body part.
Option B (ii) Pulmonary artery - takes blood from lung to heart.
Option C (iii) Aorta - takes blood from the heart to body parts.
Option D (iv) Vena cava takes - blood from body parts to the right auricle.
Q18. Identify the phase of circulation which is represented in the diagram of heart given below. Arrows indicate contraction of the chambers shown.
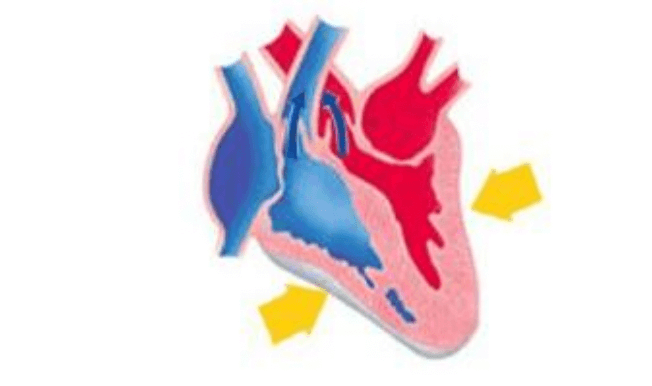
Option A Blood transferred to the right ventricle and left ventricle simultaneously.
Option B Blood is transferred to the lungs for oxygenation and is pumped into various organs simultaneously.
Option C Blood transferred to the right auricle and left auricle simultaneously.
Option D Blood is received from the lungs after oxygenation and is received from various organs of the body.
Q19. Observe the diagram of the human digestive system.
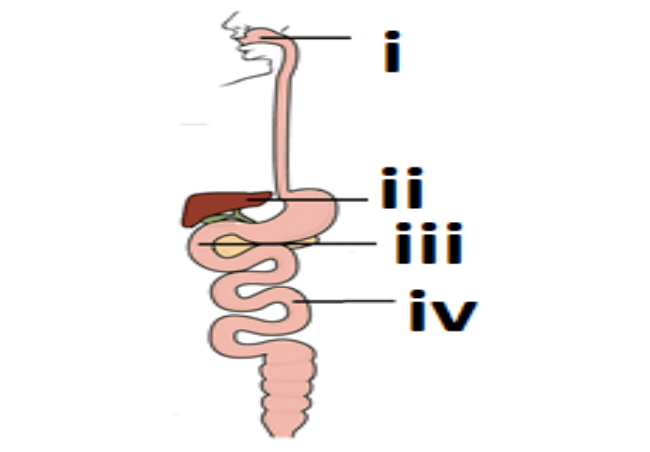
Match the labelling referred to in column I and correlate with the function in column II.
| (i) | The length of this depends on the food the organisms eat. |
| (ii) | Initial phase of starch digestion. |
| (iii) | Increase the efficiency of lipase enzyme action |
| (iv) | This is the site of the complete digestion of carbohydrates, proteins & fats |
Option A i.- a) ; ii – b) ; iii – c) ; iv- d)
Option B i.- b) ; ii – c) ; iii – d) ; iv- a)
Option C i.- b) ; ii – d) ; iii – c) ; iv- a)
Option D i.- d) ; ii – a) ; iii – b) ; iv- c)
Q20. If a beam of red light and a beam of violet light are incident at the same angle on the inclined surface of a prism from air medium and produce angles of refraction r and v respectively, which of the following is correct?
Option A r = v
Option B r > v
Option C r = 1/v
Option D r < v
Science Class 10 Term-1 Answer Key 2021 Out- Download Link
Q21. Identify Gas A in the following experiment.
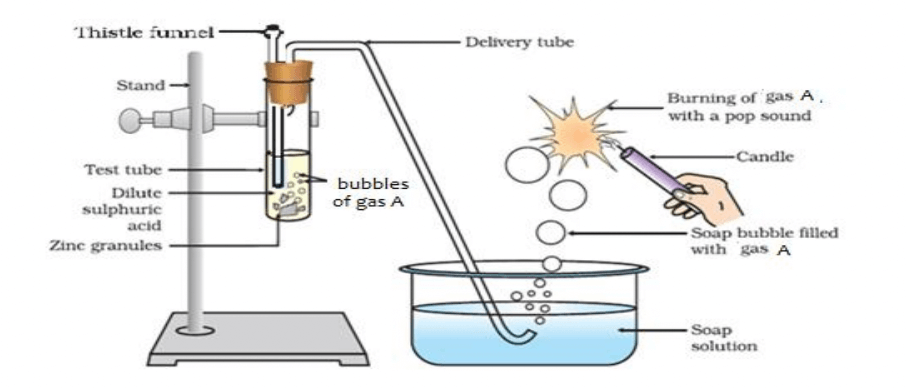
Option A Nitrogen
Option B Hydrogen
Option C Oxygen
Option D Carbon dioxide
Q22. Which diagram shows the image formation of an object on a screen by a converging lens?
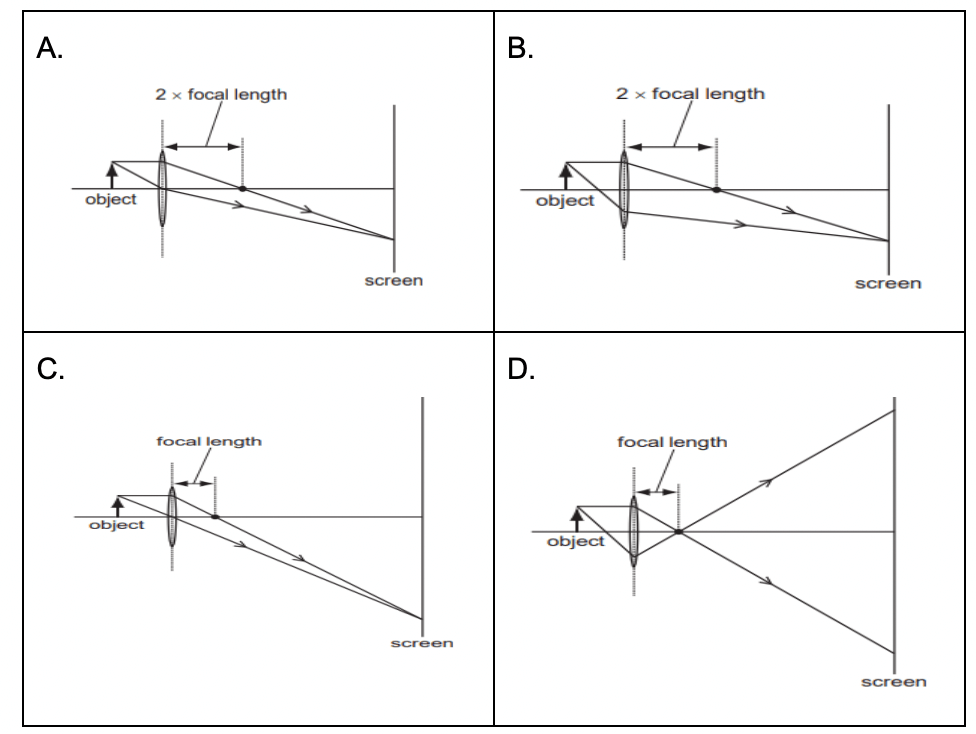
Option A a
Option B b
Option C c
Option D d
Q23. Vinay observed that the stain of curry on a white shirt becomes reddish-brown when soap is scrubbed on it, but it turns yellow again when the shirt is washed with plenty of water. What might be the reason for his observation?
i. Soap is acidic in nature
ii. Soap is basic in nature
iii. Turmeric is a natural indicator that gives a reddish tinge in bases
iv. Turmeric is a natural indicator that gives a reddish tinge in acids
Option A i and ii
Option B ii and iii
Option C i and iv
Option D ii and iv
Q24. In which of the following setups would the bulb glow?
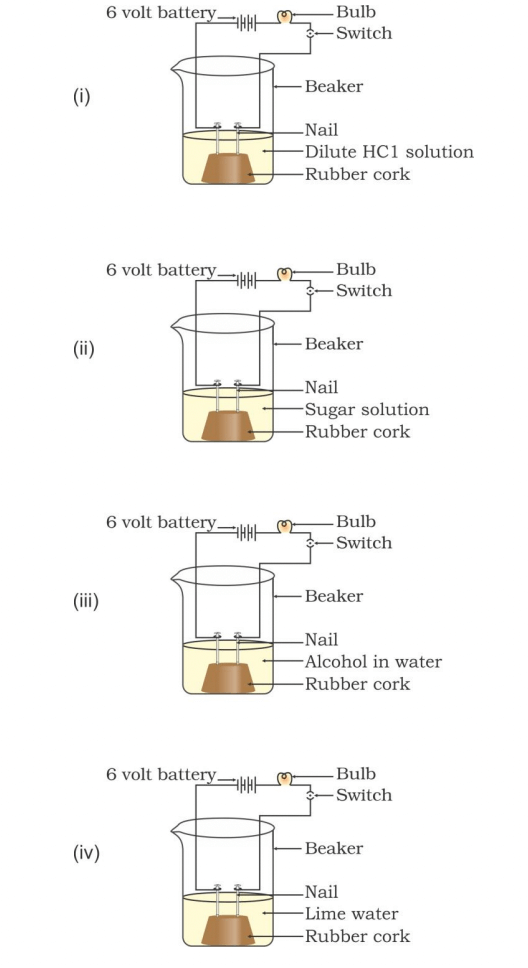
Option A i and ii
Option B i and iv
Option C ii, iii and iv
Option D i, ii and iv
Q25. The table shown below gives information about four substances: A, B, C and D.
| Substance | Melting Point | Electrical Conductivity | |
| Solid | Liquid/Aqueous | ||
| A | 295 | Good | Good |
| B | 1210 | Poor | Good |
| C | 1890 | Poor | Good |
| D | 1160 | Poor | Poor |
Identify Ionic compounds from the above-given substances.
Option A A, B
Option B B, C
Option C A, B, D
Option D A, C, D
Question No. 26 to 29 consists of two statements – Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Option A Both A and R are true and R is the correct explanation of A
Optio B Both A and R are true and R is not the correct explanation of A
Option C A is true but R is false
Option D A is False but R is true
Q26. Assertion: Fresh milk in which baking soda is added, takes a longer time to set as curd.
Reason: Baking soda decreases the pH value of fresh milk to below 6.
Q27. Assertion: Decomposition of vegetable matter into compost is an endothermic reaction.
Reason: Decomposition reaction involves the breakdown of a single reactant into simpler products.
Q28. Assertion: Resins and gums are stored in old xylem tissue in plants.
Reason: Resins and gums facilitate the transport of water molecules.
Q29. Assertion: Sky appears blue in the day time.
Reason: White light is composed of seven colours.
Q30. The table given below shows the reaction of a few elements with acids and bases to evolve Hydrogen gas
| Element | Acid | Base |
| A | ❌ | ❌ |
| B | ✔️ | ✔️ |
| C | ✔️ | ❌ |
| D | ✔️ | ✔️ |
Which of these elements form amphoteric oxides?
Option A A and D
Option B B and D
Option C A and C
Option D B and D
Q31. 5. One of the following processes does not involve a chemical reaction. That is:
Option A Melting of candle wax when heated
Option B Burning of candle wax when heated
Option C Digestion of food in our stomach
Option D Ripening of banana
Q32. It is necessary to balance a chemical equation in order to satisfy the law of:
Option A Conservation of motion
Option B Conservation of momentum
Option C Conservation of energy
Option D Conservation of mass
Q33. Rusting of iron involves a chemical reaction which is a combination of:
Option A Reduction as well as combination reactions
Option B Oxidation as well as combination reactions
Option C Reduction as well as displacement reactions
Option D Oxidation as well as displacement reactions
Q34. When ferrous sulphate is heated strongly it undergoes decomposition to form ferric oxide as a main product accompanied by a change in colour from:
Option A Blue to green.
Option B Green to blue.
Option C Green to brown.
Option D Green to yellow.
Q35. Which of the following gases is used in the storage of fat and oil containing foods for a long time?
Option A Carbon dioxide gas
Option B Nitrogen gas
Option C Oxygen gas
Option D Neon gas
Q36. In which of the following groups of organisms, blood flows through the heart only once during one cycle of passage through the body?
Option A Rabbit, Parrot, Turtle
Option B Frog, crocodile, Pigeon
Option C Whale, Labeo, Penguin
Option D Shark, dogfish, stingray
Q37. What is common between an extensive network of blood vessels around walls of alveoli and in the glomerulus of the nephron?
Option A Thick-walled arteries richly supplied with blood
Option B Thin-walled veins poorly supplied with blood
Option C Thick-walled capillaries poorly supplied with blood.
Option D Thin-walled capillaries richly supplied with blood
Q38. Plants use completely different process for excretion as compared to animals. Which one of the following processes is NOT followed by plants for excretion?
Option A They can get rid of excess water by transpiration.
Option B They selectively filter toxic substances through their leaves.
Option C Waste products are stored as resins and gums in old xylem.
Option D They excrete waste substances into the soil around them.
Q39. If the power of a lens is - 4.0 D, then it means that the lens is a
Option A concave lens of focal length -50 m
Option B convex lens of focal length +50 cm
Option C concave lens of focal length -25 cm
Option D convex lens of focal length -25 m
Q40. Rays from Sun converge at a point 15 cm in front of a concave mirror. Where should an object be placed so that size of its image is equal to the size of the object?
Option A 30 cm in front of the mirror
Option B 15 cm in front of the mirror
Option C Between 15 cm and 30 cm in front of the mirror
Option D More than 30 cm in front of the mirror
Q41. In which of the following groups of organisms, food material is broken down outside the body and then absorbed in?
Option A mushroom, green plants, amoeba
Option B yeast, mushroom, bread mould
Option C paramecium, amoeba, cuscuta
Option D cuscuta, lice, tapeworm
Q42. In a person the tubule part of the nephron is not functioning at all. What will its effect be on urine formation?
Option A The urine will not be formed.
Option B Quality and quantity of urine is unaffected.
Option C Urine is more concentrated.
Option D Urine is more diluted.
Q43. If the real image of a candle flame formed by a lens is three times the size of the flame and the distance between lens and image is 80 cm, at what distance should the candle 14 be placed from the lens?
Option A -80cm
Option B -40 cm
Option C -40/3 cm
Option D -80/3 cm
Q44. While looking at the above diagram, Nalini concluded the following
i. the image of the object will be a virtual one.
ii. the reflected ray will travel along the same path as the incident ray but in opposite direction.
iii. the image of the object will be inverted.
iv. this is a concave mirror and hence the focal length will be negative.
Which one of the above statements are correct?
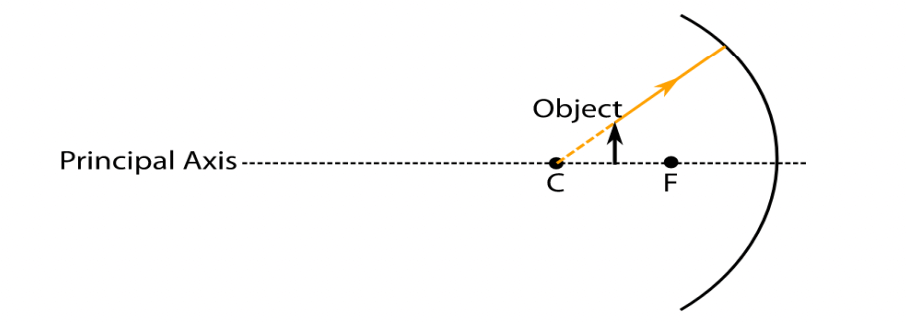
Option A i and ii
Option B i and iii
Option C ii, iii and iv
Option D i, ii, iii and iv
Q45. In the below diagram light is travelling through different media. It is noted by a scientist that ∠1= ∠3= ∠4 but ∠2 <∠1. Which of the following statement would be correct?
Option A Medium 1 is denser than medium 3 but it’s density is equal to medium 2.
Option B Medium 2 is the rarest medium.
Option C Medium 3 is denser than medium 1.
Option D Medium 1 and 3 are essentially the same medium, but medium 2 is denser than 1
and 3.
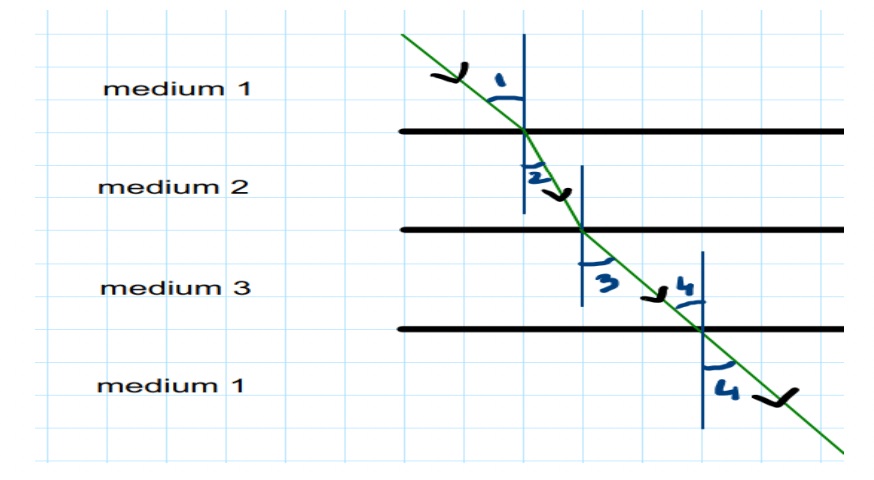
Q46. The displacement reaction between iron (III) oxide and a metal X is used for welding the rail tracks. Here X is:
Option A Copper granules
Option B Magnesium ribbon
Option C Sodium pellets
Option D Aluminium dust
Q47. The neutralization reaction between an acid and a base is a type of:
Option A Double displacement reaction
Option B Displacement reaction
Option C Addition reaction
Option D Decomposition reaction
Q48. The chemical reaction between Hydrogen sulphide and iodine to give Hydrogen iodide and sulphur is given below:
The reducing and oxidizing agents involved in this redox reaction are:
Option A Iodine and sulphur respectively
Option B Iodine and hydrogen sulphide respectively
Option C Sulphur and iodine respectively
Option D Hydrogen sulphide and sulphur
Q49. An ant’s sting can be treated with …………which will neutralise the effect of the chemical injected by the ant’s sting into our skin. Choose the correct option from the following to be filled in the blank space:
Option A Methanoic acid
Option B Formic acid
Option C Baking Soda
Option D Caustic Soda
Q50. In the reaction, identify the salt formed NH4OH (aq) + H2SO4 (aq) → _____ + 2H2O (l)
Option A NH4NO3
Option B (NH4)2SO4
Option C (NH4)3PO4
Option D (NH4)2S
Science Class 10 MCQ's Answers
The Answers to the above MCQs has been tabulated below. Cross-check your answers from here.
| Science MCQ for Class 10 Term 1- Answers | ||||
| Q1- B | Q2- B | Q3- A | Q4- C | Q5- B |
| Q6- C | Q7- D | Q8- A | Q9- C | Q10- C |
| Q11- B | Q12- D | Q13- D | Q14- C | Q15- A |
| Q16- C | Q17- D | Q18- B | Q19- B | Q20- B |
| Q21- B | Q22- C | Q23- B | Q24- B | Q25- B |
| Q26- C | Q27- D | Q28- C | Q29- B | Q30- B |
| Q31- A | Q32- D | Q33- B | Q34- C | Q35- B |
| Q36- D | Q37- D | Q38- B | Q39- C | Q40- A |
| Q41- B | Q42- D | Q43- D | Q44- C | Q45- D |
| Q46- D | Q47- A | Q48- B | Q49- C | Q50- B |
