Nitrogen is the most abundant element in the Earth’s atmosphere. The atmosphere contains almost 79% of Nitrogen. Nitrogen is present in all living organisms, in the form of protein, amino acids, and nucleic acids DNA, and RNA, while in the atmosphere it is present in molecular form (N₂) and in the form of some oxides. In its elemental form, it is a colorless and odorless gas that cannot be used by plants and animals, but after combining with Oxygen and other elements it can be used by living organisms as nutrients.
Definition of Nitrogen Cycle
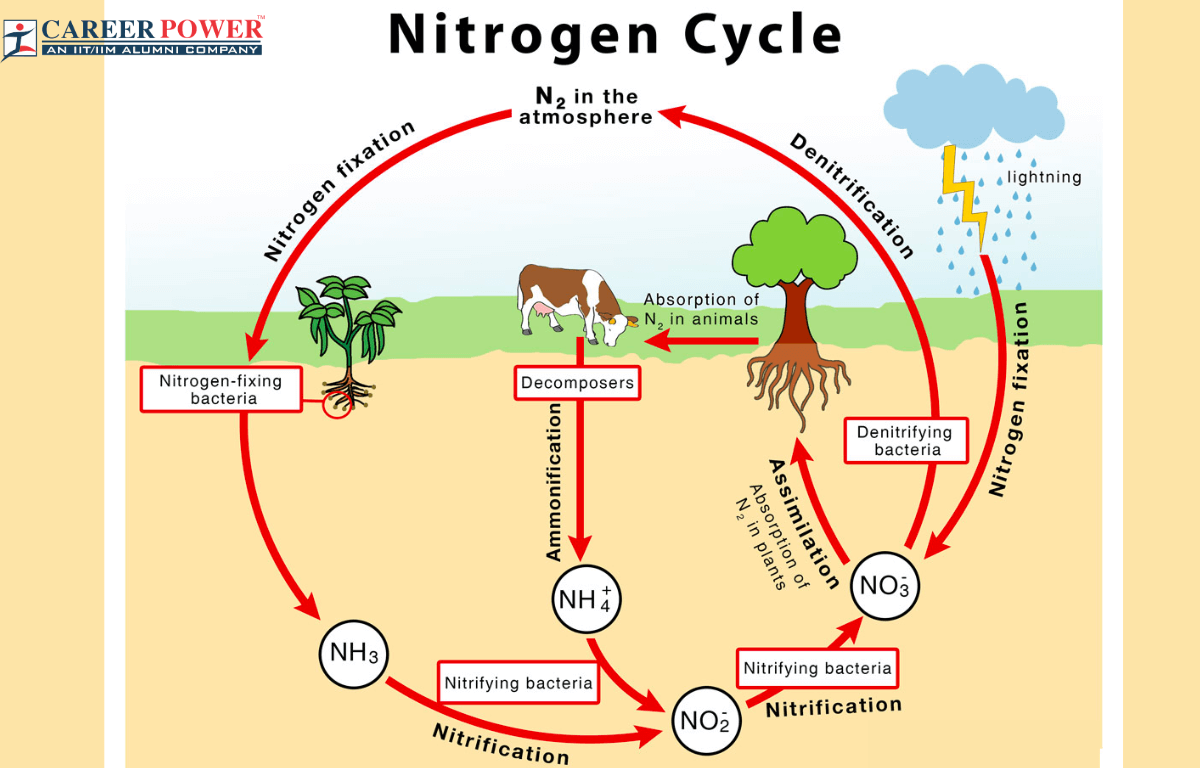
The Nitrogen cycle may be defined as the circular flow of Nitrogen from free nitrogen gas in the atmosphere to Nitrates in the soil, and then finally back to the Atmospheric Nitrogen. The atmospheric nitrogen is fixed symbiotically as well as a symbiotically by various microorganisms. Lightning and ultraviolet rays play a role in fixing a portion of atmospheric nitrogen, but the primary agents responsible for nitrogen fixation are typically nitrogen-fixing bacteria located within the root nodules of legume plants. Legumes, which are generally known for producing pulses, are the common hosts for these bacteria.
Process of Nitrogen Cycle
The Nitrogen Cycle is a part of Biology and it plays a crucial role in maintaining the balance of nitrogen in the environment and is closely related to the Carbon cycle. Part of plant proteins is consumed by animals and converted into animal proteins, while the rest is decomposed after the decay of the plant’s body, releasing nitrogen into the atmosphere.
These proteins also break into nitrogenous wastes like urea, uric acid, and ammonia in animals and are excreted out. The decomposers then act upon the dead bodies and wastes of animals releasing free nitrogen back into the atmosphere. Nitrogen is cycled from the environment to organisms and then back to the environment by several paths.
Step-Wise Process of Nitrogen Cycle
Many human activities such as the use of nitrogen-based fertilizers in agriculture and industrial processes, have significantly impacted the Nitrogen Cycle. The nitrogen cycle has many different steps that are as follows:
Step 1- Nitrogen Fixation:
Nitrogen is present in about 79% of the earth’s atmosphere, but this form of nitrogen is not directly usable by most living organisms. The reduction of atmospheric nitrogen (N₂) to ammonium ions is called Nitrogen fixation. Nitrogen fixation is the process by which certain microorganisms like cyanobacteria convert atmospheric nitrogen (N₂) into ammonia (NH3) or ammonium ions (NH4+). It is a very important step in the Nitrogen Cycle. Nitrogen-fixing bacteria are also present in the root nodules, Rhizobium bacteria, is an aerobic bacterium that needs some oxygen for its survival. To help them the leguminous plants have a hemoglobin-like protein called leg hemoglobin.
Step 2- Nitrification:
Nitrification is a two-step process in which ammonia (NH3) is firstly converted to Nitrates (NO2-) by nitrifying bacteria like Nitrosomonas and Nitrobacter. In the second step nitrate (NO₂-) is further oxidized into Nitrate (NO3-) by other nitrifying bacteria like Nitrobacter. This nitrate is then taken up by the roots as it is an essential nutrient for plants.
Step 3- Assimilation:
The nitrates and ammonia present in the soil are then taken up by the plants and primary producers and they incorporate them into their tissues to synthesize proteins, DNA, and other nitrogenous molecules. Then the consumers obtain nitrogen by consuming these plants and other animals.
Step 4- Ammonification:
When plants and animals release wastes or die the organic matter present in their tissues and wastes are broken down by decomposers into a simpler form
Step 5- Denitrification:
Denitrification is the final step of the nitrogen cycle, where nitrates (NO3-) are converted back into nitrogen gas (N₂) or nitrogen oxide (N₂O). This process takes place with the help of some decomposers like pseudomonas.
Organisms Involved in Nitrogen Cycle
The nitrogen cycle is called a “perfect cycle” in the biosphere because it maintains the overall amount of nitrogen content in the atmosphere, soil, and water. The organisms which are involved in the nitrogen cycle are mentioned below.
| Organisms Involved in Nitrogen Cycle | |
| Microorganisms | The role played in nitrogen cycles |
| Rhizobium bacteria (in the root nodules) | Nitrogen fixation (Conversion of nitrogen gas into nitrogen compound) |
| Azotobactor bacteria in soil | Nitrogen fixation |
| Blue-green algae | Nitrogen fixation |
| Putrefying bacteria | Ammonification(conversion of nitrogen-containing proteins of dead plants and animals into ammonia) |
| Fungi | Ammonification |
| Nitrifying bacteria | Nitrification(conversion of ammonia into nitrites and then into nitrates |
| Nirosomonas bacteria | Converts ammonia into nitrites |
| Nitrobactor bacteria | Converts nitrites into nitrates |
| Denitrifying bacteria (Pseudomonas) | Denitrification(conversion of nitrate salts into free nitrogen gas) |
Importance of Nitrogen Cycle
The nitrogen cycle is very important for our environment as nitrogen is also an essential element needed by plants and all living organisms in different forms.
- Essential for Living Organisms: Nitrogen is essentially required for the formation of proteins, nucleic acids, and other vital compounds in all living organisms, including plants, animals, and microorganisms.
- Plant Growth and Productivity: Nitrogen is a primary nutrient that plants need for growth and development. Plants can obtain necessary nitrogen in various forms such as nitrates and ammonium ions, which they absorb from their roots.
- Soil Fertility: The nitrogen cycle helps maintain soil fertility by maintaining the nitrogen level in it. Nitrification converts nitrogen compounds into nitrates that can be used by plants, thus increasing the fertility of the soil.
- Environmental Balance: The nitrogen cycle plays a very important role in maintaining the level of nitrogen in the earth’s atmosphere.
- Global Nutrient Cycling: The large Global Nutrient Cycle involves many essential elements like nitrogen, carbon, phosphorus, and sulfur. All these cycles are interconnected with each other and play a great role in the stability of the biosphere.











 50 Vegetables Name for Kids in English a...
50 Vegetables Name for Kids in English a...
 Food Chain: Definition, Types, Examples,...
Food Chain: Definition, Types, Examples,...
 Human Respiratory System: Definition, Di...
Human Respiratory System: Definition, Di...













