Bleaching Powder Formula: You have witnessed the term bleaching powder in many of the daily-based applications like for cleaning purposes, pure drinking water, making papers, etc. The chemical formula of bleaching powder shows that it is an inorganic chemical compound. It is to be noted here that Calcium Hypochlorite is the main component of the Bleaching Powder. Many times, calcium hypochlorite and bleaching powder are interchangeably used in day-to-day life. In this article, you will know more about the molecular formula of Bleaching powder or bleach or Calcium hypochlorite with its molecular structure, preparation of bleaching powder, properties of bleaching powder, effects of bleach, and uses of bleaching powder.
Bleaching Powder Formula
The bleaching powder formula has 2 atoms of Chlorine. Of these two chlorine atoms, one is exactly bonded with the calcium atoms. The other chlorine atom makes a bond with the oxygen atom. The chemical formula of bleaching powder is Ca(ClO)₂. In the molecular formula of bleaching powder, there is a mixture of calcium, chlorine, and oxygen atoms. The proper bond and combination results in the formation of bleaching powder. The molecular mass of bleach or bleaching powder is about 142.98 g/mol.
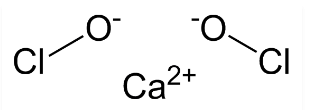
Preparation of Bleaching Powder
The bleaching powder is prepared by reacting the chlorine gas on the Calcium Hydroxide (Ca(OH)₂) or dry slaked lime. For the preparation of bleaching powder, the chlorine gas is made to produce by Chlor-alkali process and then react with dry slaked lime. The bleaching powder is also known as Calcium Hypochlorite Ca(ClO)₂ due to its molecular formula. The bleaching powder can also be treated as a soluble chemical compound that can be dissolved in plain water.
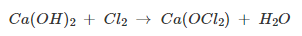
Properties of Bleaching Powder
There are different properties of Bleaching Powder as mentioned below.
- Bleaching Powder or Calcium Hypochlorite is pale yellowish in colour.
- It has a strong chlorine smell.
- It can be soluble in cold water but it is not dissolved completely. Some amount of lime available in the bleaching powder remains left undissolved.
- When bleach reacts with the strong sulphuric acid (H₂ SO₄), then the chlorine atoms available in the powder are released in the form of chlorine gas.
- It results in producing chlorine gas after reacting bleaching powder with dilute acids also.
- when the bleaching powder stays for a long period then it undergoes the oxidation process and generates calcium chlorate and calcium chloride. In this way, it decreases the bleaching effect.
Uses of Bleaching Powder
The bleaching powder or calcium hypochlorite is applied in different areas for different purposes. There are many applications of bleaching powder as explained below.
- In the textile industry, Bleaching Powder is commonly used to eliminate the colours from textile-colored items like yarn, thread, fiber, and paper through the discoloration process.
- Bleaching powder is used in the textile industry for manufacturing white clothes and apparel.
- It can kill pathogens and germs present in water and make it drinkable. So it is also used as a disinfectant for drinking water. It is believed that just a pound of bleach can make around 10,000 gallons of water pure and drinkable.
- It is considered one of the powerful oxidizing agents employed in industries. If it reacts with water then it generates strong hydrochloric acid (HCl) and atomic oxygen as a result. It is used to produce paper, textiles, detergents, and pulp in industries.
- It is also used generally at home to sanitize and clean glasses and utensils.
- It is one of the main components of Chloroform so it helps to synthesize chloroform.
- At home, it is used to wash clothes to remove dirt and stains.
- Bleaching powder is an inorganic compound that is basic in nature. It is also used as fertilizer.
Side-Effects of Bleaching Powder
Bleaching powder or calcium hypochlorite is used in many areas for different purposes. But its side effects are also present if not applied carefully. The harmful effects of bleaching powder are explained below.
- Exposure to bleaching powder in the human eye is considered dangerous. It can cause your eyes to tear up and your eyesight will be blurred or hazy.
- Bleaching powder has about 5% sodium hypochlorite which turns the pH value of bleach to about 11. So bleach causes irritation. It can cause an irritating sensation to your mouth and throat if affected.
- After applying bleaching powder while washing the hands, it is always advisable to wash it with water also. It is because too much exposure to chlorine in the human body can be devastating to the human skin.
- Over-exposure to bleaching powder can cause permanent damage to the nerve cells of the human body.
Precautions while using Bleaching Powder
For reducing the harmful effects of using bleaching powder or calcium hypochlorite, you compulsorily follow some precautionary measures as described below.
- You should not mix bleaching powder with ammonia as it will react strongly and will form a poisonous gas that can create breathing hardships.
- You should safely keep the bleaching powder on the corner so that its particles are away from your eyes and skin as it gives an irritating and burning sensation.
- In the case when you have accidentally taken the bleaching powder in your body, then you should visit the medical check-up room immediately and consult with doctors.
- In case when you accidentally inhale the bleaching powder, then it can severely damage your lungs. You should immediately visit the nearby hospital and request an oxygen support system like an oxygen cylinder, etc.
